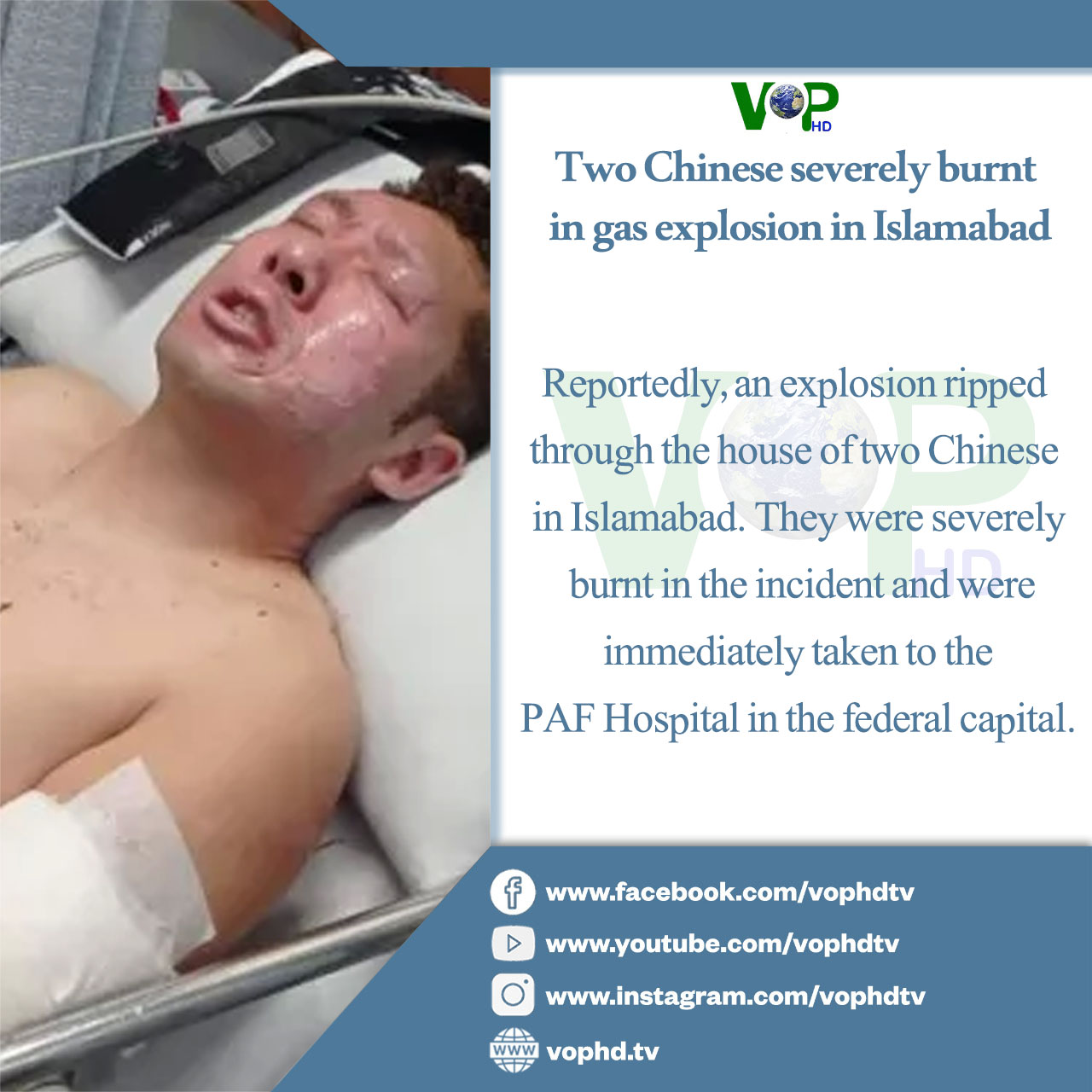
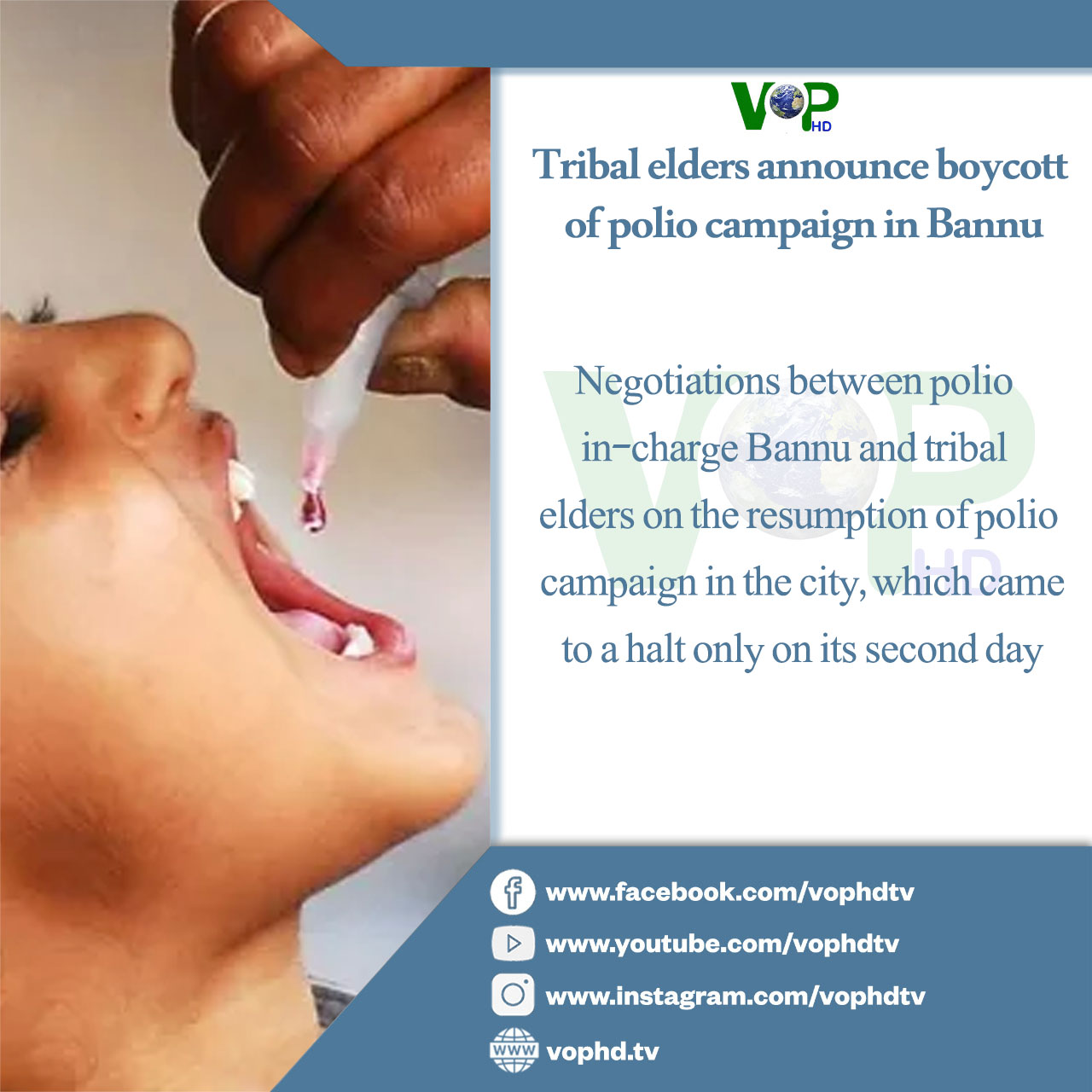
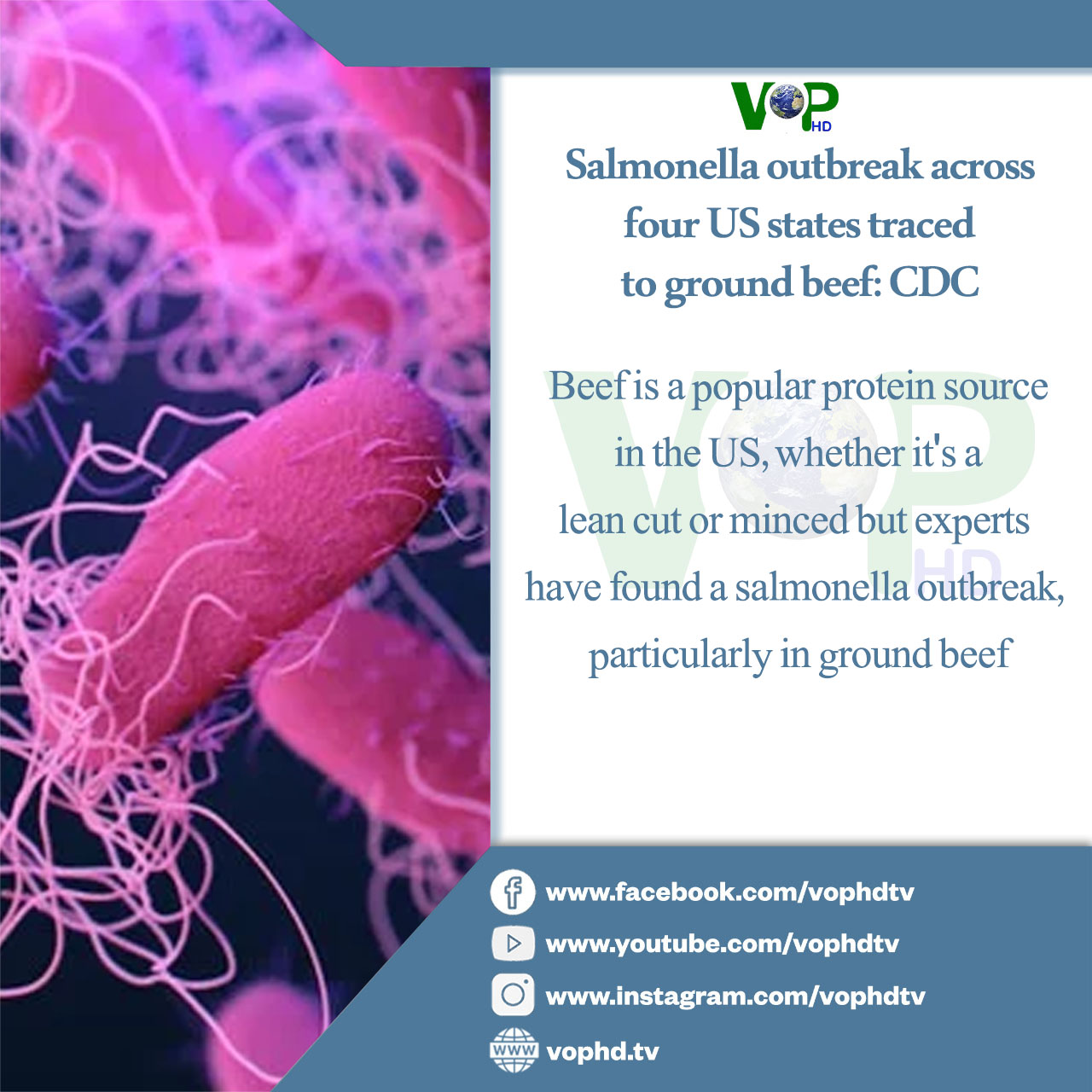
WHO probes potential links to cancer in aspartame

Researchers and healthcare organisations are examining Aspartame, a popular and widely used sweetener, to determine any potential negative effects on human health. Its possible connections with cancer are being actively sought after by experts.
Aspartame’s effects were examined last month by the International Agency for Research on Cancer (IARC) of the World Health Organisation (WHO).
The Joint Expert Committee on Food Additives and WHO is a different UN committee that is making changes to risk assessment, including the recommendation of a daily intake amount.
Chewing gum, cough drops, and toothpaste all contain artificial sweeteners in place of sugar in carbonated beverages and sodas.
Following several reviews, the US Food and Drug Administration recommended that aspartame is safe for the general public.
The IARC examined the available research to determine whether aspartame is harmful, and the Joint Expert Committee on Food Additives report is expected to provide an aspartame dosage that is safe.
The cancer research committee’s list of potential carcinogens is extensive, according to Qi Sun, an associate professor of nutrition and epidemiology at Harvard’s TH Chan School of Public Health.
Sun stated: “Consumers don’t necessarily need to be worried. Whether aspartame can be considered a carcinogen boils down to what kind of evidence we have.”
“I feel the evidence is pretty sparse to say either way to say ‘aspartame is cancerous’ or to suggest that aspartame is not as carcinogenic.”
James Farrell, a gastrointestinal oncologist at the Yale School of Medicine, said: “The people who’ve raised this question have an objective reason for raising this question. They’ve looked at it from a medical and scientific perspective … so I think it would be foolish to ignore.”
Several concerns were raised by US health officials regarding WHO having two separate reviews before the meetings.
A letter by the US Department of Health and Human Services (HHS) in August, said: “WHO’s simultaneous reviews of aspartame could potentially draw conflicting determinations that would seriously undermine confidence in the scientific process and inflame the current climate of public scepticism about the validity of science and scientific process.”
HHS stated: “The Joint Expert Committee on Food Additives should be the sole reviewer of cancer risk of aspartame in food.”
The WHO responded by saying: “The groups’ reviews would be complementary.”
“The cancer research committee, which has not previously analysed aspartame, would assess its potential cancer hazard. The food additive committee would update its risk assessment, including what it considers to be the acceptable daily intake of aspartame.”
The FDA will make its own decisions regarding aspartame in spite of experts and international organisations.
According to Professor Sun, “the agency will probably consider the evidence but is under no obligation to change its current regulations” after the reports are released in July.
The WHO issued a warning last month against using sugar substitutes to aid in weight loss because they could have long-term effects on obesity.
Sun feels that using artificial sweeteners as a temporary substitute for sugar can be a wise decision.
“I think consumers could easily switch to those household beverages to improve their health rather than get concerned about consuming artificially sweetened beverages and cancer. We don’t have evidence either way.